DISCUSSION
The data supports the hypothesis that metformin inhibits PC, however, the data does not support the hypothesis that metformin concentrations ranging from 250-500µM inhibit PC. Rather, metformin concentrations above and including 1000µM of metformin inhibited PC such that there was a significant difference between the NADH concentrations for these groups as compared to the control, according to the Duncan-Waller Post Hoc test.
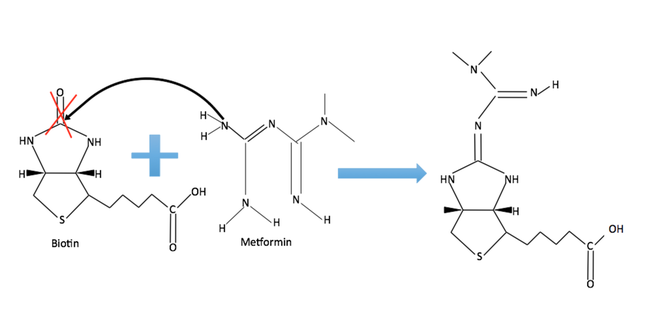
Analyzing the peaks from the 13C-NMR images reveals an interaction between metformin and biotin such that a carbonyl group normally associated with the biotin C-NMR disappeared. This suggests that metformin and biotin formed a bond when brought into contact with each other, somehow changing the structure of biotin. The proposed method of interaction is a transamination reaction, in which a nitrogen in the metformin chemical structure will attack the carbon in one of the carbonyl groups of biotin such that there will be a loss of that carbonyl group.
Analyzing the peaks from the 13C-NMR images reveals an interaction between metformin and biotin such that a carbonyl group normally associated with the biotin C-NMR disappeared. This suggests that metformin and biotin formed a bond when brought into contact with each other, somehow changing the structure of biotin. The proposed method of interaction is a transamination reaction, in which a nitrogen in the metformin chemical structure will attack the carbon in one of the carbonyl groups of biotin such that there will be a loss of that carbonyl group.
Figure 12: Proposed transamination reaction between metformin and biotin.
Biotin is a prosthetic group of PC, meaning that it will interact with PC and allow PC to function. Biotin interacts with PC through a specific carbonyl group, therefore, if metformin interacts with biotin such that the specific carbonyl group is replaced with an amine group, biotin will no longer be able to associate with PC. In this way, metformin may indirectly inhibit PC function. Although the metformin-biotin NMR did not indicate the appearance of an amine group, indicating that the proposed transamination theory is flawed, there was a loss of a carbonyl group, indicating that metformin interacted with biotin.This metformin-biotin interaction supports the metformin PC inhibition theory.
It was hypothesized that concentrations of metformin ranging from 250-500µM would best inhibit PC given that metformin is commonly found in concentrations ranging from 40-70μM in the hepatic portal vein (Duong et al., 2013; He & Wondisford, 2015). Once within the liver, the metformin concentration rises to 220 μM (Jin et al., 2009). Given this information, it was expected that lower metformin concentrations would have an inhibitory effect on PC compared to higher concentrations. Future studies should investigate even lower metformin concentrations ranging from 40-70µM to simulate hepatic portal vein conditions. Interestingly, while the 5000µM concentration group differed significantly from the control, data such as absorbance over time as well as net NADH to NAD+ conversion suggests that this high metformin concentration increased the reaction rate rather than decreased it relative to the control. Much of the controversy surrounding metformin action involves studies which use inaccurate or suprapharmacological metformin concentrations, and it may be that the anomaly of the 5000µM trend can be attributed to the excessively-high metformin concentration which may have skewed the data. It is important to note that several studies which support other metformin mechanisms of action, including through AMPK activation, also use suprapharmacological concentrations (He et al., 2009), so these data, although representative of higher than normal concentrations, still merit consideration.
Data collected from the present study may have been influenced by error and study limitations. The 5000µM and 1000µM concentration groups each had 3 trials, rather than 6 trials, and this may have impacted the distribution of error. Moreover, human inconsistencies, such as variation in starting times for the diode array spectrophotometer, possible contamination in cuvettes or in the NMR tubes, enzyme denaturation, and other such uncontrollable error may have influenced results. Although this study data can not offer definitive proof for one mechanism of metformin action over another, significant data trends can be identified in the study set which amass support for the PC inhibition theory.
It was hypothesized that concentrations of metformin ranging from 250-500µM would best inhibit PC given that metformin is commonly found in concentrations ranging from 40-70μM in the hepatic portal vein (Duong et al., 2013; He & Wondisford, 2015). Once within the liver, the metformin concentration rises to 220 μM (Jin et al., 2009). Given this information, it was expected that lower metformin concentrations would have an inhibitory effect on PC compared to higher concentrations. Future studies should investigate even lower metformin concentrations ranging from 40-70µM to simulate hepatic portal vein conditions. Interestingly, while the 5000µM concentration group differed significantly from the control, data such as absorbance over time as well as net NADH to NAD+ conversion suggests that this high metformin concentration increased the reaction rate rather than decreased it relative to the control. Much of the controversy surrounding metformin action involves studies which use inaccurate or suprapharmacological metformin concentrations, and it may be that the anomaly of the 5000µM trend can be attributed to the excessively-high metformin concentration which may have skewed the data. It is important to note that several studies which support other metformin mechanisms of action, including through AMPK activation, also use suprapharmacological concentrations (He et al., 2009), so these data, although representative of higher than normal concentrations, still merit consideration.
Data collected from the present study may have been influenced by error and study limitations. The 5000µM and 1000µM concentration groups each had 3 trials, rather than 6 trials, and this may have impacted the distribution of error. Moreover, human inconsistencies, such as variation in starting times for the diode array spectrophotometer, possible contamination in cuvettes or in the NMR tubes, enzyme denaturation, and other such uncontrollable error may have influenced results. Although this study data can not offer definitive proof for one mechanism of metformin action over another, significant data trends can be identified in the study set which amass support for the PC inhibition theory.